- Boron nitride ceramic structure
There are three variants: hexagonal, hexagonal close-packed, and cubic.Hexagonal crystal BN is a stable phase at normal pressure;close-packed hexagonal crystal BN and cubic crystal BN are stable phases at high pressure, and metastable phases at normal pressure;
hexagonal crystal boron nitride transforms into cubic crystal nitrogen at high temperature and high-pressure boron nitride or close-packed hexagonal crystal boron nitride.
Boron Nitride Ceramics
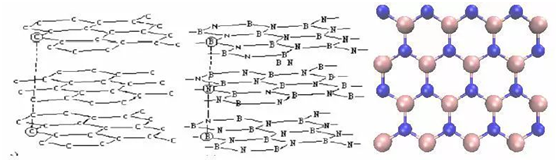
Hexagonal boron nitride Ceramic
Hexagonal crystal boron nitride has a layered structure similar to graphite, its color is white, and its performance has many similarities, so it is also called “white graphite”.
- The manufacturing process of boron nitride ceramics
(1) Preparation of boron nitride powder:
① boron halide method
This method is also called the gas-phase synthesis method. The boron halide reacts with ammonia to form an intermediate amino complex, and then the intermediate is processed at a high temperature to obtain BN. At 125~130°C, the intermediate object decomposes into B2(NH)3, and when it is heated to 900~1200°C, B2(NH)3 is decomposed into BN.
② Boron anhydride method
By reacting boron anhydride with ammonia at high temperatures,
By reacting boric anhydride with sodium cyanide (calcium),
By reducing boron anhydride with graphite in a graphite crucible
③ Borax method
Borax-ammonium chloride method: Na2B4O7+2NH4Cl+2NH3—4BN+2NaCl+7H2O
Borax-urea method: Na2B4O7+2(NH2)2CO—4BN+Na2O+4H2O+2CO2
(2) Forming of boron nitride ceramics
In order to obtain dense boron nitride, hot pressing is mainly used. Boron nitride ceramics can also be formed by various molding methods according to different shapes and sizes.
(3) Sintering of boron nitride ceramics
In order to facilitate the sintering of boron nitride, a certain amount of additives can be added. Commonly used additives include B203, Si3N4, AIPO4, BaCO3, etc.
The amount of B2O3 added to the additive should be appropriate. The presence of B2O3 will cause the boron nitride ceramics to absorb moisture and lead to a sharp deterioration in electrical and thermal properties;
If BaCO3 is added, the performance of hot-pressed boron nitride ceramic products can be improved.
- Properties and applications of Hexagonal boron nitride Ceramic
| Properties | Applications |
| White in color, stable in performance, and good in machinability | used as a high-temperature lubricant |
| high-temperature resistance and insulation | used as an electrical insulation material |
| Thermal conductivity and penetrating performance to microwave radiation | used as a transmission window for radar in the electronics industry |
|
High melting point, small thermal expansion coefficient, and stable to almost all molten metals, |
used as a high-temperature metal smelting crucible, heat-resistant material |
| Used as a heat sink and heat conduction material (temperature up to 2800°C in the neutral or reducing atmosphere) | |
| used to make the separation ring of the crystallizer in the billet horizontal continuous casting technology in the metallurgical industry | |
| The best material for manufacturing engine parts, widely used in aerospace |

Manufacturing and Application of Boron Nitride Ceramics
Declaration: This is an original article of INNOVACERA®. Please indicate the source link when reprinting: https://www.innovacera.com/news/manufacturing-and-application-of-boron-nitride-ceramics.html.




 Enquiry
Enquiry